News
Article
Understand the 2025-2026 Flu Vaccine Composition Recommendations
Author(s):
Key Takeaways
- The 2025-26 influenza vaccine composition was determined by an internal panel, differing from previous years' external expert involvement.
- The primary update for the 2025-26 vaccine is the inclusion of a new influenza A/H2N2 strain, while maintaining a trivalent formulation.
The only major change is in the included strain of influenza A/H2N2 virus, which has been updated for both the egg-based and the cell- and recombinant-based vaccines.
The FDA has released its influenza vaccine composition for the 2025-26 influenza season in the US. This year, the FDA’s recommendation for strains to be included in the vaccine came from an internal panel from the FDA, the CDC, and the Department of Defense.1 This differs from previous years, with an external panel of experts contributing to the decision-making process in a meeting held March 13, 2025.2 Thirteen of this year’s panelists had a PhD. Of note, only 4 panel members have a health care degree, all physicians, but not all are currently practicing physicians. The vaccine recommendations must be made early in each calendar year because it takes months to prepare the vaccines for use in the fall.
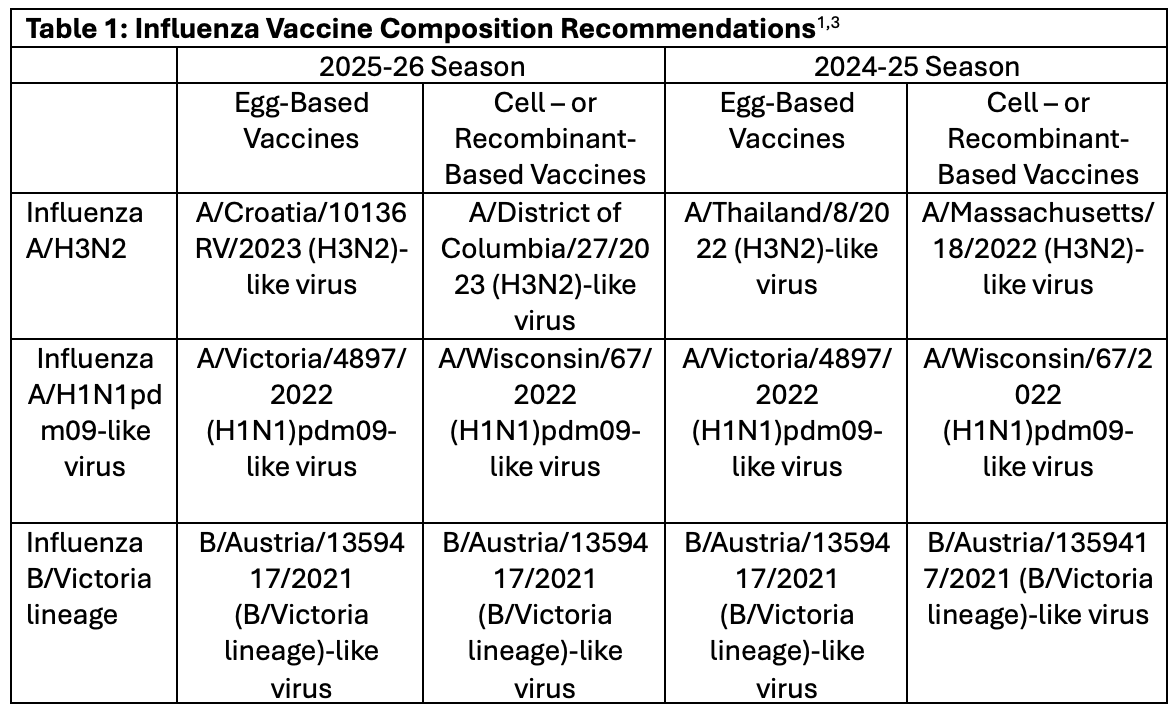
For the 2025-26 influenza season, there are very few changes from the 2024-25 season (Table 1).1,3 In fact, the only major change is in the included strain of influenza A/H2N2 virus, which has been updated for both the egg-based and the cell- and recombinant-based vaccines. Vaccines will continue to be trivalent. Strains of influenza B/Yamagata lineage have not been known to circulate in multiple years and are not considered to be a public health concern.3 According to data presented during the meeting, a global analysis of circulating influenza strains demonstrated that the influenza A/H1N1pdm09-like virus and influenza B/Victoria lineage virus recognized the specific subclades, or subtypes, of those viruses well enough to provide protection. On the other hand, multiple substitutions on the hemagglutinin antigen of the influenza A/H3N3 emerged, resulting in a reduced reactivity of the current season’s components against some circulating strains. Tests of the influenza A/H3N2 strains, which are now included in the recommended components for the 2025-26 season, appear to recognize the majority of circulating influenza A/H3N2 well, which prompted the panel to support the shift to their inclusion.
Table 1: Influenza Vaccine Composition Recommendations1,3
The 2024-25 influenza season has been the most active among all age groups since 2017-18 and the hospitalization rate is the highest since 2010-11.4 Approximately 94.7% of influenza this season has been caused by influenza A, with an almost even distribution of those between H1N1pdm09 (50.8%) and H3N2 (49%). Approximately 99.4% of the influenza A/H1N1pdm09 viruses were well-recognized by the 2024-25 included strain. In contrast, only 54.9% of influenza A/H3N2 viruses were recognized by this year’s included strain of that virus. All of the influenza B/Victoria-lineage viruses were well recognized by the vaccine strain.
Image credit: Leigh Prather | stock.adobe.com

It is important to remember that this is laboratory data, and data analyzing actual vaccine effectiveness at preventing influenza infection in the general population will not be available for some months after the current season comes to a close. It is also essential to understand that although vaccination against a virus like influenza, where it is impossible to predict with 100% certainty which viruses will circulate, the vaccine is still considered the best way to prevent influenza. Over the past 10 years, vaccine efficacy has ranged from as low as 19% to as high as 60%.5 Midseason data from the Department of Defense regarding effectiveness in 2024-25 has ranged from 40% to 57%.6 This is higher than the 35% effectiveness that experts predicted before the start of the season, similar to the 42% seen during 2023-24, and higher than the 34.5% effectiveness seen specifically in the Southern Hemisphere in the 2023-24 season.5
Even when effectiveness is low, vaccination make a big clinical impact. Patients who are vaccinated are more likely to have a milder infection, less likely to have influenza-related complications, less likely to be hospitalized, and less likely to be admitted to the intensive care unit.5 This is the greatest benefit of the influenza vaccine, particularly for patients who are vulnerable to influenza-related complications including older adults, children younger than 2 years old, and patients with co-occurring conditions.
As the very busy influenza season starts to end for health care professionals, it can seem overwhelming to start preparing for the next season. Vaccination remains a key tool in reducing the impact of influenza on public health, preventing severe illness, and serious complications. Health care providers should continue to advocate for the importance of annual vaccination and help patients understand its role.
References
1. Influenza Vaccine Composition for the 2025-2026 US Influenza Season. FDA. Updated March 13, 2025. Accessed March 29, 2025. https://www.fda.gov/vaccines-blood-biologics/influenza-vaccine-composition-2025-2026-us-influenza-season
2. Interagency Meeting to Discuss and Make Recommendations on the Strain Composition of Influenza Virus Vaccines for Use in United States During the 2025-2026 Influenza Season. FDA. March 13, 2025. Accessed March 29, 2025. https://www.fda.gov/media/185810/download?attachment
3. Use of Trivalent Influenza Vaccines for the 2024-2025 US Influenza Season. FDA. Updated March 5, 2024. Accessed March 29, 2025. https://www.fda.gov/vaccines-blood-biologics/lot-release/use-trivalent-influenza-vaccines-2024-2025-us-influenza-season
4. Weekly US Influenza Surveillance Report: Key Updates for Week 9, ending March 1, 2025. CDC. March 7, 2025. Accessed March 29, 2025.https://www.cdc.gov/fluview/surveillance/2025-week-09.html
5. 2024-2025 Flu Vaccine Effectiveness: What Do We Know? Flu.com. Updated September 2024. Accessed March 29, 2025. https://www.flu.com/Articles/2024/2024-2025-Flu-Vaccine-Effectiveness
6. Department of Defense Influenza Surveillance and Mid-Season Vaccine Effectiveness. March 13, 2025. Accessed March 29, 2025. https://www.fda.gov/media/185795/download?attachment
Newsletter
Stay informed on drug updates, treatment guidelines, and pharmacy practice trends—subscribe to Pharmacy Times for weekly clinical insights.